The U.S. drug regulator on Friday greenlighted the use of coronavirus vaccines developed by Pfizer Inc. and Moderna Inc. for booster shots for all adults, expanding the eligibility from older and higher-risk individuals.
Authorizing the use of a booster dose for individuals 18 years of age and older helps to provide "continued protection against COVID-19, including the serious consequences that can occur, such as hospitalization and death," Acting Food and Drug Commissioner Janet Woodcock said in a press release.
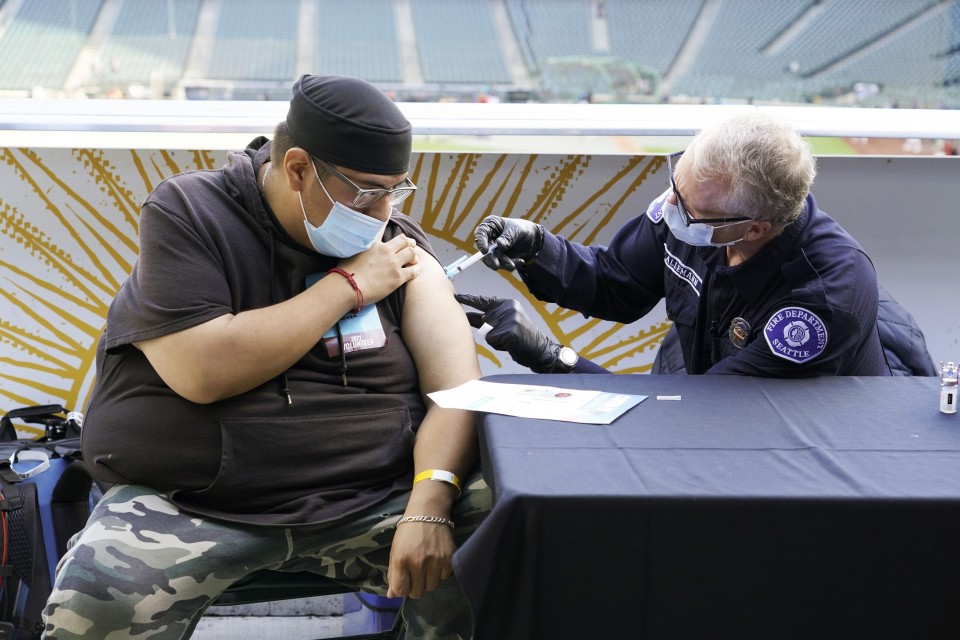
Up to now, booster shots were only allowed for individuals 65 years of age and older, individuals 18 through 64 years of age at high risk of severe COVID-19, and those of the same age group with frequent occupational exposure to the virus.
Under the latest authorization, any adult would be eligible for a single booster dose of either the Pfizer or Moderna vaccine at least six months after their second dose of a two-shot inoculation, or at least two months after receiving Johnson & Johnson's single-dose vaccine.
Booster shots have been rolled out even as more than 40 percent of the U.S. population has yet to be fully vaccinated, according to data by the Centers for Disease Control and Prevention.